If 61.5 L of oxygen at and an absolute pressure of 2.45 atm are compressed to 38.8 L and at the same time the temperature is raised to , what will the new pressure be?
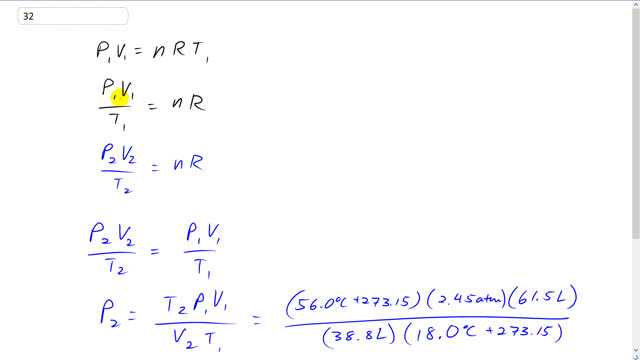
In order to watch this solution you need to have a subscription.
This is Giancoli Answers with Mr. Dychko. When we write the ideal gas law, we have to look at what's constant and what's changing between the two scenarios before and after the temperature is raised and the compression happens and so on. So, initially we have a pressure of 2.45 atmospheres. And then there's gonna be a new pressure after the compression. So, that means the pressure is gonna change. And so in the first scenario we'll give it a subscript 1 and in the second scenario we'll give it a subscript 2. And the volume also is gonna change. So, it needs a subscript V1 here and V2 down here. The number of moles of oxygen is not going to change because the container is sealed. So, n does not need a subscript and R is just the universal gas constant. And the temperature does change so, it means at T1 here. So, we have P V over T1 by dividing both sides by T1 equals n R. And in the second scenario after the compression, we'll have a new pressure and a new volume and a new temperature and P2 V2 over T2 is also also going to equal the same n R. So, since they both equal n R, they are equal to each other. And then we can solve for P2. Multiplying both sides here by T2 over V2 and we get pressure 2 is temperature 2 times pressure 1 volume 1 divided by volume 2 times temperature 1. So, temperatures are in absolute, temperature is kelvin. The pressures, we can leave as atmospheres because, well, if we put our pressure in atmospheres here, that means our answer will also be in atmospheres. The volume, we can leave as liters because the liters are just going to cancel. And you could convert them into cubic meters if you like but it doesn't make any difference because the conversion factor will cancel anyway. The temperature you always have to convert it into kelvin because the conversion is not created by multiplying by a conversion factor that it's going to cancel, instead the conversion involves adding a number and so this does not cancel, the 273.15 does not cancel on the top and bottom because it's not a factor, it's a number that's added, it's a term. So, you have to do that conversion. Well, add 56 degrees Celsius as the final temperature plus 273.15 times 2.45 atmospheres times 61.5 liters to begin with divided by the final volume of 38.8 liters after it's compressed times by the initial temperature of 18 degrees Celsius plus 273.15. And you get 4.39 atmospheres will be the pressure after it's been compressed.
