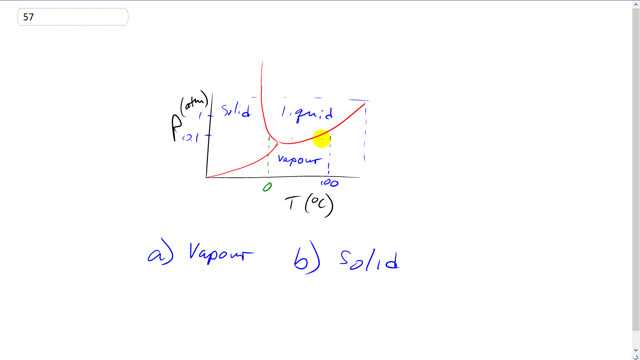
Water is in which phase when the pressure is 0.01 atm and the temperature is
- vapour
- solid
In order to watch this solution you need to have a subscription.
This is Giancoli Answers with Mr. Dychko. This is the phase diagram for water, and it looks like at 0.1 atmospheres at 90 degrees Celsius, looks like it's just below this curve here, which means it is a vapor, it's not going to be liquid at 90. At sea level pressure 1 atmosphere, it is liquid, that would be here and that point were it crosses 1 atmosphere in 90 degrees. But as we have it here with 0.1 atmospheres, looks like that corresponds to about there this point here, 90 degrees 0.1 atmospheres, that is in the vapor region. And then part b, at negative 20 degrees Celsius it's going to be definitely solid because here 0 degrees Celsius and negative 28 degrees Celsius puts this about here or so.
